What we learned building the Gold Cohort—and how it’s shaping Phase 2 of the Marathon of Hope Cancer Centres Network

By Dr. Jim Woodgett, President and Scientific Director, Terry Fox Research Institute, and Dr. André Veillette, Executive Director, Marathon of Hope Cancer Centres Network
In 2019, the Government of Canada made a landmark investment to accelerate precision oncology and transform how cancer is studied and treated across the country. That investment launched Phase 1 of the Marathon of Hope Cancer Centres Network (MOHCCN), a bold, pan-Canadian initiative led by the Terry Fox Research Institute and the Terry Fox Foundation.
From the outset, our ambition was clear: to unite Canada’s cancer research and clinical communities around a shared vision and, together, create a national strategy to accelerate precision oncology research and to bring it to more patients, regardless of where they live.
Five years later, that vision has taken tangible form.
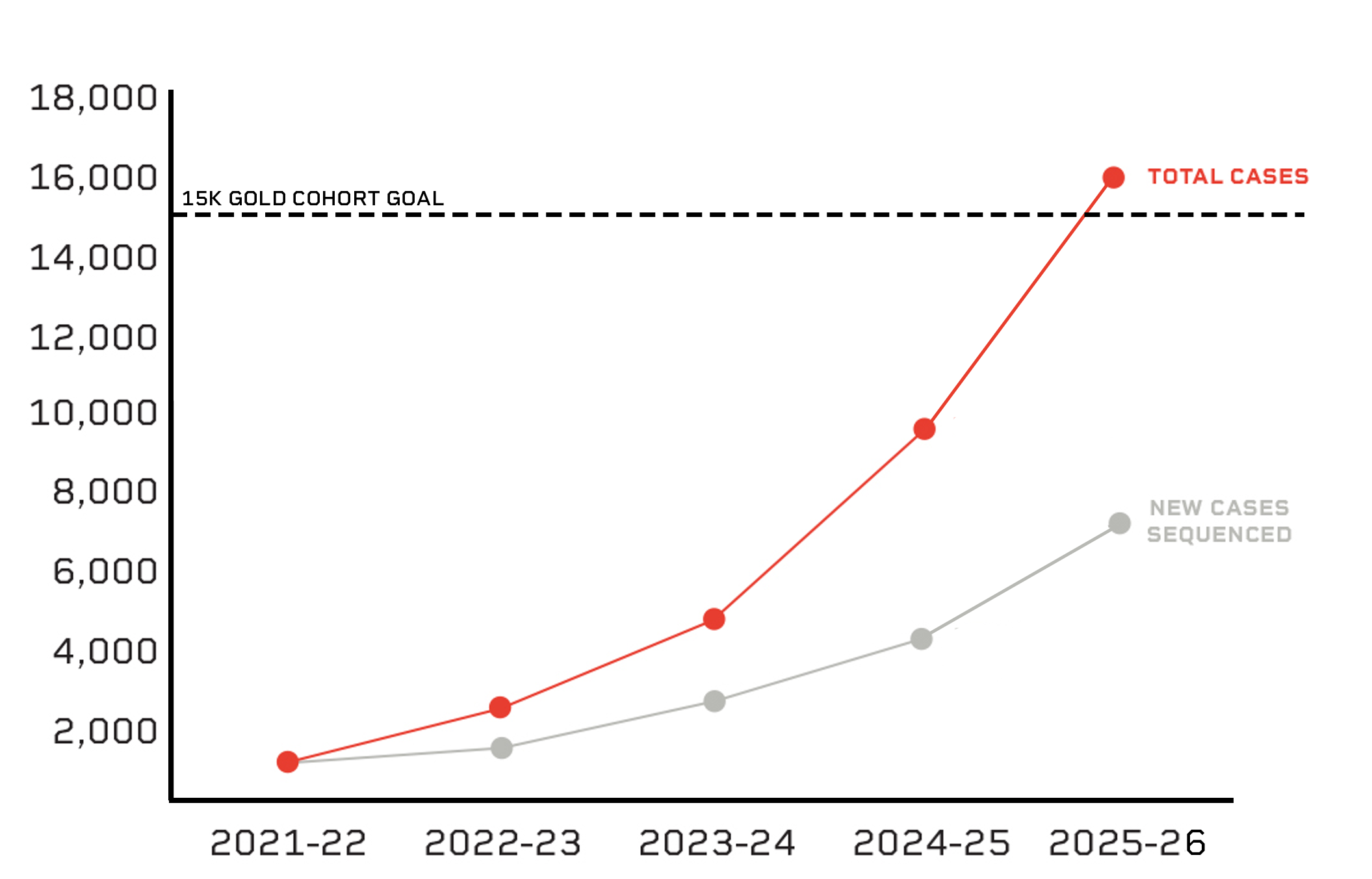
More than 1,300 collaborators across 54 institutions and over 100 partners have come together to build a truly national effort. Among its most significant achievements is the creation of the MOHCCN Gold Cohort, Canada’s largest and most comprehensive cancer case resource, integrating clinical and genomic data from more than 15,000 patients.
This is a foundational asset for the country. But building it was not straightforward.
As we conclude Phase 1, it is worth reflecting on the lessons learned over the last five years, and how they’re shaping our approach to Phase 2 of the Network.
Building a national network takes time…and trust
Although funding for the Network was announced in early 2019, operations formally began in mid-2021, following the signing of a Contribution Agreement with Health Canada. Those intervening years were shaped by the COVID-19 pandemic, which disrupted research, delayed activities and challenged institutions across the country.
Despite this protracted process, progress did not stall. Researchers, clinicians and leaders continued to meet, plan and align priorities. By the time the Network officially launched, a shared roadmap was already in place.
Even then, early progress was slower than we hoped.
Looking back, this should not have come as a surprise. Canada had never attempted to harmonize genomic and clinical data collection across institutions at this scale. Each centre had its own infrastructure, workflows and levels of readiness. Building a unified system required not only technical alignment, but trust between researchers, oncologists and administrators, who were all working at different institutions, subjected to different regulations.
The establishment of working groups made up of experts from across the country proved essential to building trust and working through these challenges. These groups created policies, defined standards and – perhaps most importantly – helped bridge differences in practice. By sharing knowledge, experience and resources, we were able to set up a collaborative platform where larger centres supported smaller ones and capacity was built across the country.
The impact of this collaborative model and the framework it helped establish became clear over time. After a slow start, data generation accelerated significantly, with the number of fully profiled cases nearly doubling year over year. What began as a complex coordination challenge evolved into a scalable national system.
Clinical data: essential, complex, and resource-intensive
One of the key differentiators of the Gold Cohort is that every case in the resource includes well-annotated clinical data that pair with the genomic data. This will allow researchers to study how genomic features influence things such as diagnosis, treatment response, disease progression and patient outcomes—information needed to translate molecular insights into meaningful improvements in patient care.
But if generating genomic data at scale across the country presented technical challenges, clinical data revealed a different kind of complexity.
Capturing treatment histories, outcomes and patient trajectories requires time, expertise and careful validation. This is especially the case in a federated healthcare environment like Canada’s, where healthcare systems do not capture clinical data in a uniform way. Overcoming these challenges took both time and many discussions, but eventually a standardized clinical data model was created, and centres across the country were able to implement data curation protocols that aligned with their own systems, allowing for standardized data to be collected across the country.
Creating these protocols took time, which meant that clinical data collection lagged that of genomic data. This was, at least in part, by design. Centres typically confirm that genomic data meet quality standards before investing in clinical annotation. In addition, meaningful clinical data often depend on events that occur after sample collection, such as treatment response.
Through this process, one lesson became clear: high-quality clinical data is both indispensable and resource-intensive, meaning expensive in effort and time.
Phase 1 highlighted the need to explore new approaches, including the partial automation of data extraction and curation. As we move into Phase 2, improving the efficiency and scalability of clinical data ingestion will be critical to maximizing the value of the Gold Cohort, as will reducing costs of these processes.
Flexibility as a necessity
This leads to one of the most important lessons we learned during Phase 1: that flexibility is essential when operating at scale.
The Gold Cohort was built on rigorous standards, but applying those standards in real-world settings required thoughtful adaptation. A clear example emerged during the Network’s “Year of Data Acceleration” in 2023–2024.
Many centres reported cases where whole-genome sequencing (DNA) was successful, but transcriptome sequencing (RNA) did not meet quality thresholds, particularly in biobanked samples, where RNA degradation is common. Maintaining a strict requirement for both would have excluded valuable data.
The solution was to evolve the definition of a Gold Cohort case. Tier A cases include both DNA and RNA profiling, while Tier B cases include DNA-only data. This adjustment preserved data quality while expanding the utility of the dataset. Today, approximately 85 per cent of cases include both data types.
This principle extends beyond genomics.
Clinical data models, for example, proved more difficult to standardize across all cancer types. Blood cancers, in particular, present unique characteristics that do not always align with solid tumour frameworks. Rather than forcing uniformity, the Network adapted, refining data fields and planning further enhancements for Phase 2.
Flexibility, in this context, is not about lowering standards. It is about ensuring that standards remain relevant, inclusive and scientifically meaningful.
Representation matters
Beyond the quality of the data, a central goal of the Network was to create a data resource that reflects the diversity of Canada’s population and enables advances in precision oncology that will benefit all patients, not only those commonly represented in this type of research.
Phase 1 made important progress in this direction. The Network expanded to all ten provinces in just three years, bringing together institutions from coast to coast. The Canadian Spectrum Working Group also established guidelines to identify underserved communities and populations underrepresented in cancer research.
However, it is clear to us that important gaps remain.
Participation from the Territories has been limited, and representation of some groups – including First Nations, Inuit and Métis communities – has likely fallen short of demographic expectations. In many cases, this has been compounded by the lack of consistent socio-demographic data, making it hard to know precisely how representative of the full spectrum of Canada's diverse populations the Gold Cohort really is.
This is a critical area for improvement.
Phase 2 includes a concerted effort to expand into rural, remote and northern regions, and to strengthen partnerships with underserved communities. The development of a new socio-demographic data model will also support more ethical, consistent and meaningful data collection, helping ensure that the benefits of precision oncology are more equitably distributed.
Strong foundations enable future adaptation
Scientific research rarely unfolds exactly as planned. Technologies evolve, knowledge advances and new questions emerge.
That’s why the Network has remained open to exploring new profiling technologies that could be incorporated into the Gold Cohort in future phases of the Network. The exploration of these new technologies, supported through the TFRI-MOHCCN Technology Development Awards, is yielding exciting insights that will help us expand the potential for impact of the Gold Cohort in Phase 2 and maintain the Network at the forefront of innovation.
Although we’ve continuously adapted to the reality on the ground and explored new profiling technologies to get a more complete picture of cancer, one of the most striking aspects of the MOHCCN is how closely it has remained aligned with its original vision. The core principles established at the outset – collaboration, excellence, national coordination and patient impact – have endured, and so has our capacity to expand the potential for impact of the Gold Cohort.
This is not by chance.
These principles were shaped through broad consultation, informed by international experience and continuously refined through input from experts across and beyond the Network. That strong foundation allowed the Network to adapt operationally without losing sight of its goals.
Looking at the future
Since operations officially began in 2021, we have:
United more than 1,300 patients, researchers, clinicians, administrators and donors from over 100 institutions across the country to form the Team Canada of Cancer Research...
Created Canada’s largest and most complete cancer case resource...
Funded impactful cancer research across the country...
Bolstered Canada’s human resource capacity for clinician scientists, health informaticians and data scientists...
Elevated the voice of patients in research...
And helped bring precision oncology to more Canadians!
With the Network now firmly established, the focus will shift to expanding both scale and scope—reaching more patients and embedding genomic analysis more deeply into clinical care in a cost efficient and time efficient manner. Just as importantly, it will test how this infrastructure performs in real-world settings, where its impact on patient outcomes can be fully realized. It reflects the extraordinary efforts of a national community united by a shared purpose. More importantly, it provides a foundation for the next phase of discovery, innovation, and impact.
If Phase 1 was about building the system, Phase 2 is about testing it—at scale, across the country and for the benefit of all Canadians.
As we conclude Phase 1, it is worth reflecting on the lessons learned over the last five years, and how they’re shaping our approach to Phase 2 of the Network.
Related Team Members
-
Jim
MOHCCN Network CouncilHeadquarters and OperationsWorking Group Member
Woodgett -
André
Executive Director
Veillette
